
- Details
- Written by: Developer SM
- Category: Perspectives
- Hits: 58

- Details
- Written by: Developer SM
- Category: Perspectives
- Hits: 68
We live in a world inundated by metrics and governed by KPIs. Even medicine, with its patient-centricity, is often clouded by the need to achieve.
The race toward being the first to discover the cure for X disease has overshadowed much of the original purpose of modern medicine: to actually make a difference. Accolades and awards are nice, but what really matters is how the patients receiving these treatments respond to them. What are their experiences? How does drug discovery and innovation benefit their daily lives on a real and practical basis? Not every avenue of medicine cares about that but Jonas Hannestad, Gain’s new Chief Medical Officer, does.
It’s clear from the moment you meet Dr. Hannestad that empathy is his MO. His warm and inviting smile is unexpected but welcome, especially in the fight for the future of neurodegenerative disease drug discovery. One might expect Hannestad to be all business, and while he’s certainly focused on getting results, his approach sees a bigger picture.
Hannestad cares. His smile shows experience and understanding. Hannestad comes from a blended background of academia and biotech, the combination of which has been instrumental in his ability to translate theoretical concepts into practical applications. Many researchers in biopharma are solely focused on treating the targeted symptoms that define certain neurodegenerative diseases. For example, memory loss with Alzheimer’s disease. And while that is a critical component of developing impactful drugs, there’s another aspect that often gets left by the wayside: quality of life.
How people live with these diseases matters just as much, and Hannestad understands that. Receiving FDA clearance and approval will always be paramount, but how you receive approval can’t be at the expense of the patient experience. As we sat down to discuss his new role as Chief Medical Officer of Gain, Hannestad explained how he arrived in this position. Passionate, eloquent, and driven to make a difference, he is dedicated to not just hitting numbers but really enacting meaningful change for the patients living with neurodegenerative diseases like Alzheimer’s, Parkinson’s, and ALS.
_________________________________________________________________________
You’ve had a multitude of experiences throughout your career. Not only have you worked across multiple areas of disease, but you’ve also held different roles in various biotech and Pharma (SVP of Clinical Development at Alkahest, Medical Director at Denali Therapeutics, and Director of Neuroscience Discovery Medicine at UCB Pharma).
How have your past experiences helped you develop your current perspective on drug development?
Well, working across several therapeutic areas, not just within neurology and neurodegeneration but also outside of CNS, has helped me understand the foundation of drug development and the commonalities that exist regardless of the specific area of drug development. I find that very helpful because you learn from each experience you have, and over time these diverse experiences coalesce into something larger, and you can apply that learning moving forward.
For instance, say I learn something from an ophthalmology program. I may be able to apply some of that to a Parkinson’s program at a later time. I would say the same is true, but in a different way, for my academic work, which largely focused on basic physiological mechanisms that are common to multiple diseases. Drawing such parallels across diseases and contexts has been very helpful for the roles I’ve held in biotech and pharma. I’ve always been in that early clinical development/ translational space where you have to understand how the mechanism and the target pathways fit with your disease of interest and how you can demonstrate that in a clinical trial.
Translational medicine was a primary focus of your time in academia. What have been some major influences from your time in the corporate world thus far?
I built my foundation of translational medicine in my academic work, both in basic cell and molecular biology research and then in clinical research. I was then able to take that knowledge base (CNS pathophysiology) and put it to work in the corporate world where the focus is more narrow. Rather than trying to understand the biology underlying a disease process, in biotech you’re evaluating whether a molecule and a mechanism could one day become a new medicine for a certain group of patients. It’s much more focused than academic research, and I fell in love with that as soon as I had my first drug development experience.
Would you say one has been more rewarding than another?
I would, yes. If I had to pick one, I would say that the biotech drug development aspect is more rewarding to me than academic work because it’s more tangible. You can actually see how this could lead to something that could help somebody. Although there are a lot of failures, obviously, there is a chance that the drug you’re working on could lead to a new treatment one day, whereas the academic work is often very removed, very basic, very theoretical. It’s very interesting intellectually, but it’s not as tangible.
Do you think you would be able to appreciate the value of your work in biotech quite as much without the foundational background of academia?
I probably would be able to appreciate it, but I don’t think I would be as good at it. If I’d gone directly from my clinical training into industry, I may not have been able to contribute as much.
Here’s how I see it: I think that if you “grow up” in the pharma industry by going directly into the workforce right after your studies, you learn a very specific, a very practical way of thinking and working.Whereas if you have prior academic experience, I think it broadens your view and your thinking so you can apply it in a different way.
That’s incredibly valuable, that broader perspective. You’ve certainly had the opportunity to broaden your scope, having worked across a number of neurodegenerative diseases, namely, Parkinson’s, Alzheimer’s and ALS. Can you speak to how you’ve seen the field change? Are there any similarities, any areas of overlap, and any stark differences in the treatment methods for these diseases?
One of the biggest changes that has occurred in the last 10 to 20 years across all 3 of those neurodegenerative diseases is our knowledge of the genetics and the mutations that cause or contribute to diseases. It’s not just the mutations that directly cause the familial forms of these diseases, but also the genetic variants that increase your risk of developing one of these diseases. These are very important, because those mutations in Alzheimer’s, Parkinson’s, and ALS have increased our understanding of the pathophysiology by pointing to specific pathways that are contributing to the onset and progression of these diseases
So many of these mutations are not necessarily a starting place for a treatment. They could be targeted directly in some cases, but not always. Still, they point you to certain mechanisms that may be relevant and that can be targeted in other ways.
And the interesting thing is that there are a lot of commonalities across those 3 diseases, which 30 years ago, people wouldn’t have thought because they present so differently. They affect different areas of your brain and the symptoms that accompany these diseases are very different, so the symptomatic treatments are very different. Symptomatic treatments for Parkinson’s have nothing to do with the symptomatic treatment for Alzheimer’s, for example. But if you look closely at the underlying molecular biology, which I think has been informed by those genetic discoveries, you realize that there’s a lot in common in terms of mechanisms such as protein synthesis and quality control, mitochondrial function, lysosomal function, autophagy, and inflammation.
Would you say there are similarities in the approach to drug discovery for these diseases?
Yes, that, too. In my experience, drug discovery efforts for these diseases were based on animal models with face validity (phenotypically similar to the human disease), but this approach didn’t work well. But now animal models are largely used to mimic the molecular biology events that we know occur in the human disease process, more than the phenotype. I think this approach will prove more successful.
So that’s one thing. Then the other is in the early clinical development space, where the focus has shifted from the clinical or approvable endpoints to the biomarkers that measure effects of the investigational drug on the targets and mechanisms. The focus is to basically show that your molecule does what it was designed to do in humans.
Our goal is to show that the biology fits and the that the drug actually works in the same way in humans as it does in mice and rats.
This leads well into the discussion of the rapidly changing field of drug discovery. How do you stay up-to-date with the latest developments? How do you decipher between what’s just “buzz-worthy” and what will actually make a difference?
That’s very challenging, especially in early research. When somebody identifies a new potential target or mechanism in one of these diseases, it’s hard to predict whether that will eventually have an impact on a patient with that disease. You have to just follow the field and understand what the people who have been working on the disease for a long time think.
The most relevant thing for me is making sure that we’re developing drugs not just to gain FDA approval but to also treat aspects of the disease that are important to patients. Because those two don’t always match. A lot of the clinical endpoints that have been developed in these diseases have been developed primarily by clinicians who treat the patients. And yes, those clinicians know a lot about the disease, but it’s from the clinician’s perspective. They have not usually lived the experience. Sometimes what a clinician focuses on are the certain aspects of the disease that are easier to observe or measure in the doctor’s office, but those may not be the most important aspects to the patients themselves. There’s been a lot of work on this over at least the last decade. When you ask patients what’s most important to them, it sometimes surprises you as a clinician.
Do you have any specific examples?
Yes. Sticking with Parkinson’s, for example. Parkinson’s is primarily known as a disease characterized by certain motor components; tremors, bradykinesia (slowness of movement), muscle stiffness, and impaired gait and balance.Those are the 4 key components that were originally described by Parkinson and are still used to diagnose the disease today.
But there are a lot of other symptoms that go along with Parkinson’s, like depression and anxiety, cognitive dysfunction, constipation, problems with blood pressure regulation, urinary function, erectile function, etc. that were not part of that initial description, and they’re not typically used for diagnosis, but they’re obviously very important to patients. So the symptomatic treatments for Parkinson’s, the dopaminergic treatments, while they treat the motor symptoms fairly well, they often do not improve those other symptoms that are not part of the core syndrome.
So if somebody has mild depression that goes along with Parkinson’s, they may be less likely to exercise, which is something that’s been shown to be very beneficial in Parkinson’s. Or they may be less likely to stick to taking their medications on a certain schedule. It all comes together. So it’s really important to not just address the symptoms that are direct indicators of the disease but also the accompanying symptoms that may seem to be indirectly related, but are just as important for patients.
Now with all of this a solid groundwork, we pivot to your introduction as Gain’s Chief Medical Officer. What are you most excited about in terms of joining Gain Therapeutics?
I think I’m most excited about the fact that Gain is on the verge of going into patient studies. Taking a new compound from preclinical testing into healthy volunteers is a very important step in drug development, but what makes me most excited is addressing the question: Does this drug have the potential to help people with a certain condition or disease?
That’s the stage where Gain is; we’re about to go into patient studies, and that’s very exciting.
In addition, I’m very confident in this mechanism, the development of the drugs that target the molecular pathways affected by GBA mutations. These mutations are associated with an increased risk of Parkinson’s, and the underlying molecular mechanisms make a lot of sense. It has a lot of support. To your earlier question regarding how you know if something’s going to be relevant for patients or not; you don’t really know, nobody really knows, but you can have more or less confidence, I suppose, and for me, having GBA as a target is something in which I have more confidence than some of the other targets. After all, it’s hard to work on something that you don’t really believe in, right?
In your opinion, what sets Gain apart from other drug discovery companies?
I think many biotech companies and pharmaceutical companies have a lot in common just by the nature of the industry. The way drugs are developed confines you, and things have to be done in a certain way; some of those things simply can’t change and they’re going to be the same across the board. That’s a good thing, having commonalities because they’re foundational to how we determine whether drugs are safe and effective.
But I think what makes Gain different from some of the other companies that are currently working on Parkinson’s is that it’s not just the target, the GBA mutation target. There are a few other companies working on that, but it’s the specific way in which this molecule was designed by Gain that works in a slightly different way than the compounds from some of the other companies in this space. Other companies that develop drugs for this target are developing molecules that activate the enzyme, whereas the Gain molecule helps the enzyme move along inside the cell in a way that sort of prevents the sort of clogging up of cellular systems that happens in people with this mutation.
So what you’re saying is, Gain isn’t just building a better mousetrap, so to speak; there are actual functional differentiators with the Gain molecule.
Yes; the data that we have in both cell systems and in mouse/rat studies indicates that this molecule works in a different way than other molecules that are purportedly for the same mutation.
What do you anticipate for the future of drug discovery, either within Gain or within the industry as a whole, for the next 5 to 10 years?
The thing that I’m most excited about is that the field has made a lot of progress in understanding the basic physiology of these diseases. But we’re still pretty far from having it figured out. Specifically, we’re pretty far from where other disease areas are. So I’m hoping that the field will continue to progress so we can understand it to such an extent that we can be more confident in developing new drugs.
Trying to come up with a treatment for a disease that you don’t really understand is like a bit of a shot in the dark, and unfortunately that’s kind of what we’ve been doing for the past 40 or so years. That’s why there have been so many failures. So I’m hoping that will start to shift and that the increased understanding will lead us to more confident targets. Right now we’re at a point where we can often only say that there’s statistically about a 10% chance that something will work. I want to get to a point where it’s a 40 – 50% chance because once we do, we’ll develop many, many more new treatments for these diseases. So that’s something I’m very hopeful for in the next 5 to 10 years.
You mentioned a desire to see treatments for these neurodegenerative diseases get to a point where they’re as advanced as some of the others. Which others are you referring to?
In terms of treatments, some areas in which we more or less understand how they work are cardiovascular disease, oncology, metabolic disease, autoimmunity – those are probably the main ones.
So you’re saying the neurodegenerative space is looking to hit the benchmarks those fields hit. How do you feel about the current pace of progression? Do you think things are going at a quicker pace than they used to?
It’s hard to say if it’s going at a quicker pace, but it’s definitely going forward. It’s kind of like 2 steps forward and 1 step back. For example, there was recently an ALS drug in phase 3 that didn’t work. They had gotten accelerated approval based on promising phase 2 data, but then they did phase 3 and it didn’t work out. They had to pull it off the market, which was a good decision, because you don’t want a drug out there that doesn’t work and gives patients false hope. But it’s the same for the anti-amyloid therapies for Alzheimer’s. These treatments probably have some effect, but it’s a very small effect and it’s not to the degree that we’re looking for. So it’s a little bit of progress, but not as much as we want.
Would you say that the progress that it offers is more impactful or more insightful than it has been in the past?
Yes, I think so. In the past, in Alzheimer’s specifically, there were only symptomatic treatments that didn’t have any effect on disease progression. These amyloid treatments, you know Eisai’s drug, and probably Lilly’s drug [donanemab], if it gets approved, they do appear to slow progression. It’s not just symptomatic treatment. Now while that effect on slowing disease progression is very small, if it’s real it means we’ve made progress. We know that we’re on the right path. Or one right path.
Yes. Even if you’re not all the way there, you’re inching toward something that could really make a huge difference. And essentially, that’s what you’re looking to do.
There’s something happening here. The progress may be slow, relatively speaking, but every step is forward-moving and that’s what matters.

- Details
- Written by: Developer SM
- Category: Perspectives
- Hits: 145

- Details
- Written by: Developer SM
- Category: Perspectives
- Hits: 54

- Details
- Written by: Dhaval Gupta
- Category: Perspectives
- Hits: 81

- Details
- Written by: Dhaval Gupta
- Category: Perspectives
- Hits: 81

- Details
- Written by: Dhaval Gupta
- Category: Perspectives
- Hits: 56
Professor Taylor highlights the groundbreaking work of Gain Therapeutics in developing GT2287, a GBA modulator, as a potential treatment for Parkinson's disease. She discusses the origin of GT2287, its mechanism of action, and preclinical findings demonstrating its efficacy in addressing key pathological features of Parkinson's disease. For a comprehensive understanding of the subject matter, we recommend watching the video for an in-depth analysis. Alternatively, to grasp the key themes discussed, peruse the accompanying blog for an overview.
Introduction to Gain Therapeutics' GT2287
Hartaj Singh: Great, Tiago. Thank you so much. I appreciate you going over so much in-depth material, and especially focusing on molecular biology and pathology. I think Dr Alcalay and you have brought us to a point where we can now hand it over to Gain’s own Dr Joanne Taylor. She is the Senior Vice President of Research here and she's going to discuss the recent in vivo data for GT 02287. It’s a best-in-class small molecule for the treatment of GBA Parkinson's disease and other neurodegenerative diseases. So Joanne, please take it away.
GT2287: Discovery and Clinical Development
Professor Taylor: Thank you very much, Hartaj. And thanks very much to our previous two speakers who have undoubtedly made my job a lot easier. So as Hartaj said, I'm going to introduce you to our GBA modulator, GT2287, which is currently in the clinic as a treatment for GBA1, Parkinson's disease.

So this molecule was discovered from our proprietary computational drug discovery platform. Actually, all of the molecules which were in our pipeline were discovered employing this platform. So we're delighted that GT 2287 is now in the clinic as proof of concept of the utility of this platform. And before I get going, I'd like to acknowledge the generous support of various foundations that have financially contributed to this work over the years. They include the Michael J. Fox Foundation, the Silver Star Foundation, [indiscernible] and Eurostars.
The Role of GT2287 in Parkinson’s Disease

So I don't think I need to go through this slide at all, given the two excellent previous presentations, just to reiterate that we think that restoring GCase function is expected to stop or slow the progression of Parkinson's disease. And we're hoping that that's what our candidate molecule GT 2287 is going to be able to do.
Efficacy of GT2287 in Cell-Based Experiments and Animal Models

So we believe that GT 2287 addresses the primary path of physiological effects in the disease cascade resulting from GK’s function that we've heard quite a lot about already from our previous two speakers. And in a plethora of cell-based experiments, some of them using cells derived from patients, and also animal models in which we have modelled GBA1 Parkinson's disease, we have seen that our compound can ameliorate lots of the effects of GCase dysfunction. So, for example, we've been able to see effects on endoplasmic reticulum stress, which is caused when misfolded GCase is not able to traffic through the cell to the lysosome where it needs to be to perform its function. This causes an accumulation of toxic lipids, that substrate of the enzyme as well as Alpha-synuclein accumulation, and we've also been able to show in many, many experiments that we can prevent this from happening with our compound.
The lysosomal dysfunction that ensues from all of this buildup of Alpha-synuclein and lipids, and GCase not being where it's supposed to be, we can also stop this happening with our compound and we can also stop the accumulation of mitochondrial reactive oxygen species. It's become clear recently that not only is GCase important in the lysosome, but it needs to get to the mitochondria as well where it's important in energy homeostasis. So having this functional GK seems to affect mitochondria, which we know are very, very important in neurodegenerative diseases, including Parkinson's disease.
And then in animal models, we've been able to show that our compound affects neuroinflammation, that it prevents neuronal cell death, particularly dopaminergic neuronal cell death, and it prevents the ensuing motor deficits from all this cascade of pathology. So because of this, we expect GT 2287 to have potential as a disease-modifying therapy in Parkinson's disease, as well as other neurodegenerative diseases where a deficit in GCase function has been implicated. So we think that our compound has a best-in-class profile of the GCase targeting drug candidates that we know about to date. It's orally available, it's a brain-penetrant small molecule, and it's an allosteric GCase regulator; it binds away from the active site, so it doesn't interfere with sub-straight processing. And by the mechanism that we heard about earlier, this compound chaperones GCase to the lysosome, where it needs to be to perform its function. And as I already mentioned, this compound is already in Phase 1 clinical trials.
GT2287’s Advantages and Allosteric Regulation
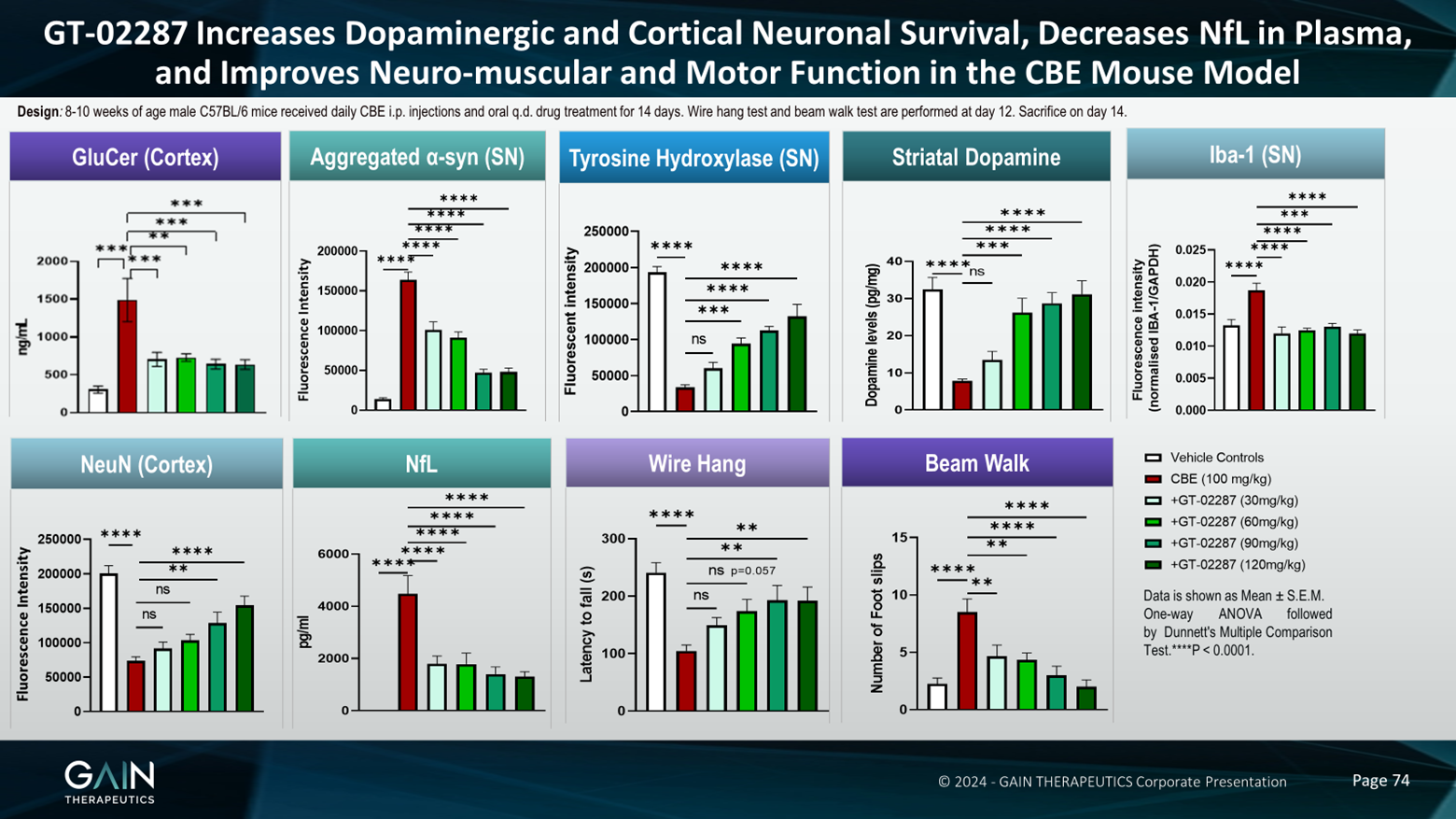
So I'd like to present to you some data in a model which we've used a lot to test this compound and also follow on compounds. It's a model where we've tried to model GBA1 Parkinson's disease in normal animals. So the way we've done this is if we used a compound called CBE, which is an irreversible inhibitor of the GCase enzyme, we administer this compound to the animals and this takes out some of the activity of the GCase enzyme, and it's irreversible. So we can forget that GK is in the model, we've taken it out of function, and then we are testing our compound on the effect on the remaining GCase, which hasn't been inhibited by this compound. So we are trying to model the reduction in GCase activity that we see in GBA1 Parkinson's disease.
In-depth Look at GT2287’s Performance in Animal Studies
And what we see in this model, and what we see with our compound is everything that you'd like to see, really, in a model of GBA1 Parkinson's disease. And with our compound, we see everything that you'd like to see with a molecule, which is treating this disease. So, for example, we see an increase in glucosylceramide, which is a toxic lipid substrate, and we're able to prevent this with our molecule. We've heard a lot about aggregated Alpha-synuclein in this meeting, and in our model, we see a big increase in aggregated Alpha-synuclein and we're able to prevent this with our molecule.
Likewise, we see a decrease in the number of dopaminergic neurons. We see this with tyrosine hydroxylase staining. Tyrosine hydroxylase is an enzyme important in the metabolism or the making of dopamine, and we're able to prevent the loss of these dopaminergic neurons. Maybe unsurprisingly, because we're losing dopaminergic neurons, we also lose striatal dopamine, which we can prevent with our molecule.
We see an increase in neuroinflammation. We see this with EB1 staining. It's a marker of microgliosis. Again, we prevent it with our molecules. We're also preventing the loss of neurons in the cortex. The cortex is, of course, also very important in Parkinson's disease and we've heard, we see cognitive decline, especially in GBA1 Parkinson's disease. So it's important that we can rescue these neurons in the cortex as well.
Very exciting. In our model, we see an increase in the neurofilament light chain in the plasma. So neurofilament light chain is emerging as a marker of neurodegeneration. The fact that we can see it in the plasma suggests that this could be a biomarker for the effects of our compound in an accessible body fluid, i.e., the plasma. The brain is not accessible and there are some issues with taking cerebral spinal fluid as well.
Evaluating GT2287’s Effectiveness After Disease Onset
And then just to round this off, as you might expect, given the hypothesis of how this all works, and given all the other effects that we've seen, we're able to rescue the motor deficits that we see in this model. So when we measure the amount of time a mouse can hang on to a wire or an upturned cage lid, when we administer the CBE, the latency to fall, that's the red bar, is very much reduced. And then we bring this back up towards control levels with our compound. And then in a beam walk experiment, which is measuring coordination, so we count the number of foot slips that the animals make, they're much less coordinated in this model and we're able to prevent this with our compound.
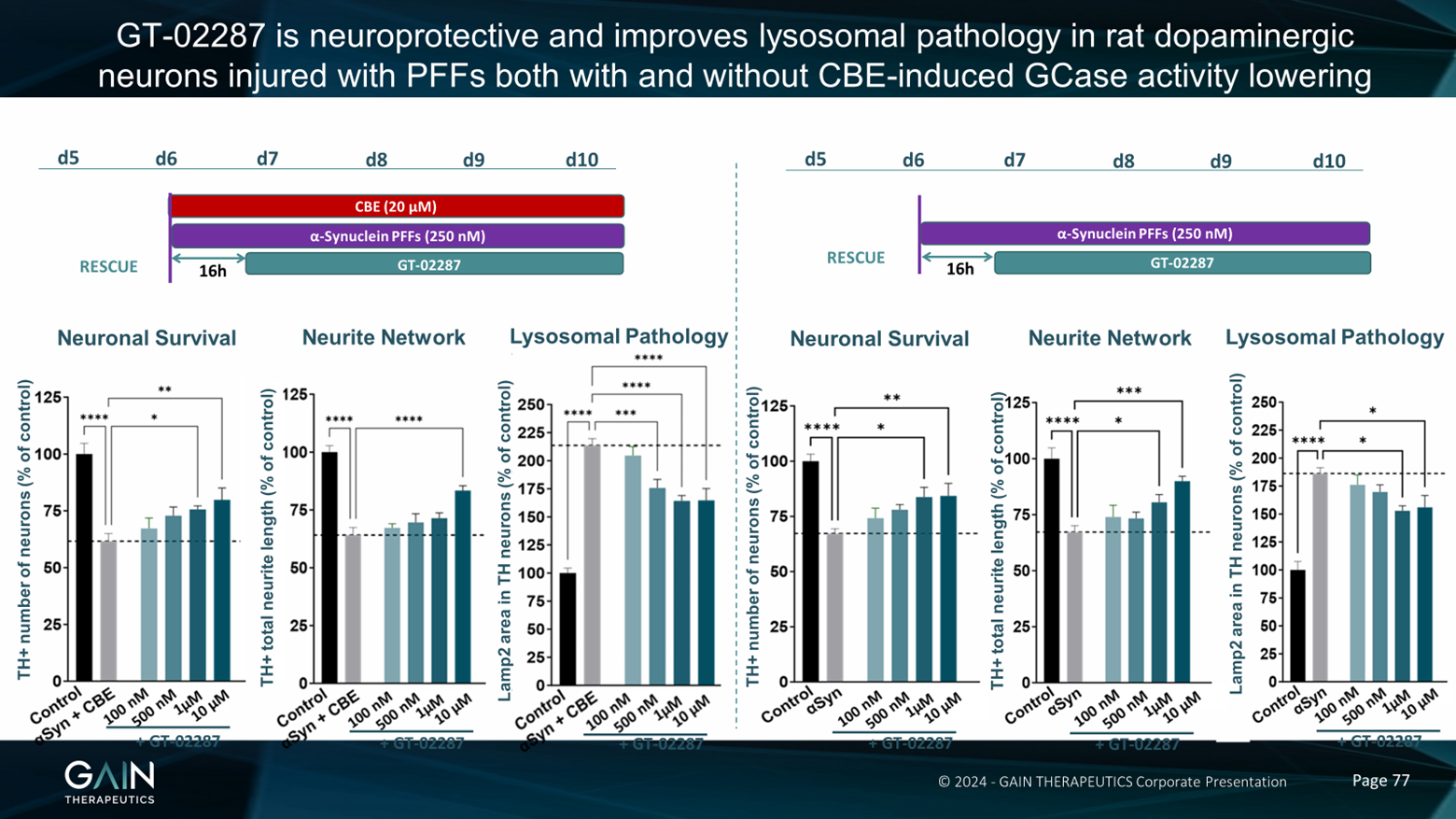
Phase 1 Clinical Trials and Future Plans

Now, all of the experiments that I just described to you were done by giving our compound at the same time when we introduced the toxic insult. So the really important thing to know is, does our molecule work when the disease process is already underway? So we wanted to take a look at this. And, first of all, we took a look at this in cultured dopamine neurons. So we were looking at the effect of our compound in cultured neurons in a dish. And we've cultured these neurons, and then, as I've just described to you, in the animal model, we've added CBE to these neurons, again, to knock down some of the GCase activity. And then in addition, in this model, we've added Alpha-synuclein-preformed fibrils. So as we heard from Dr. Outeiro when the Alpha-synuclein aggregates together, then this becomes more kind of toxic and pathological, we've added this into the culture as well. And we can see comparing the black control bars and the grey bars, that when we add CBE and Alpha-synuclein, we get a reduction in neuronal survival, and we get a reduction in the neural network. And we also get an increase in lysosomal pathology.
Now, when we add our compound after the start of the experiment, so when all the disease processes are in place, on the next day, essentially, we're still able to rescue these effects on neuronal survival network and lysosome or pathology. What's very interesting, given the discussion that we've just had about idiopathic versus GBA1 Parkinson's disease, is we see very, very similar results when we treat these neurons not with CBE, so we're not affecting the GCase, but when we just add Alpha-synuclein, preformed fibrils, and we see very similar effects in the cell-based model, and we see very similar effects of our compound, i.e., we can rescue this effect and prevent these neurons from dying even in a delayed treatment paradigm.
So we've now taken this into animals, this therapeutic paradigm. And again, we've given the animal CBE, but this time quite a low amount of CBE; half the dose that we gave in the previous in vivo experiment I described to you and we're only giving it every other day. And as we have done with the cell culture experiments, we've also administered Alpha-synuclein, performed the fibrils and we've injected these straight into the brain. Again, to mimic what happens in Parkinson's disease and specifically in this experiment GBA1 Parkinson's disease.
And then once we've got this whole disease cascade going, we started to treat these animals with our compound after either a four-day delay, or an eight-day delay, and then we measured the motor function of these animals in a wire hang test, which I described to you before. So we tested the animals on day 14 of the experiment, which is about a week after we started treating with the compound, and on day 27 of the experiment, which is about three weeks after we started treating with the compound. Then again, we did the motor performance, and the wire hang, and then we also measured plasma NFL as a measure of neurodegeneration.
Conclusion and Acknowledgments
So as I mentioned, this compound is already in the clinic. It's in a Phase 1 trial in healthy volunteers. The upcoming milestones for this trial are this quarter, we expect to complete the single ascending dose part of the phase one trial and to start the multiple ascending dose part of this trial. In the coming quarter, we expect to finish the med part of the trial and we also expect to be able to collect some data looking for effects on GCase activity and levels even in these healthy volunteers. I may not have mentioned but I should have mentioned that our compound seems to act on non-mutated GCase in terms of helping it get to the lysosome and increasing activity as it does in mutated GCase. So we want to look at GCase activity and levels in healthy volunteers as a first inhuman proof of concept.
And then in the second half of this year, spanning out to the first half of next year, we want to add a patient cohort onto the Phase 1 trial, where we will start to look at biomarkers that you can measure then in patients. So, for example, neurofilament light chain, given what we've seen in our preclinical models and other markers of, say, for example, neuroinflammation and so on, and how these lipid substrates are moving as well. And then later on, we expect to have a US IND filing, and then to start our Phase 1B/Phase 2 clinical trial.
So I don't have time for the conclusions, actually, but I think we can go to the question and answer session. But just before I finish, I'd like to point out that I didn't do all this work and it's been carried out by past and present members of the Gain team over the years. I would particularly like to call out Dr Beatrice Guzman, who has produced all of the in vivo data that I shared with you today, and Dr Natalia Perez, who provided the cell-based data.

- Details
- Written by: Developer SM
- Category: Perspectives
- Hits: 517
What Makes a Truly Valuable AI Drug Discovery Platform?
The value in AI drug discovery platforms lies in their ability to target known, yet previously unreachable, therapeutic targets. Gain Therapeutics’ Magellan™ exemplifies this by combining AI with a physics-based approach to identify novel allosteric binding sites on disease-implicated proteins.

- Details
- Written by: Developer SM
- Category: Perspectives
- Hits: 206
Gain Therapeutics is ready to share the latest R&D update on Parkinson’s Disease. This event is intended for all KOL’s, Analysts, Patient Advocates, and investors who share a common interest in neurodegenerative diseases.
“Beyond Symptomatic Treatment of #Parkinson’s Disease: Gain Therapeutics R&D Update with KOL & Analyst Insights 2024”
This R&D update event will occur Thursday, February 22nd, 2024 between 11:00 am – 12:00 pm EST.
Registration information can be found here: https://lnkd.in/erx-c4vn
Participants are encouraged to send questions to
Discussion Topics will include:

- Details
- Written by: Developer SM
- Category: Perspectives
- Hits: 350
Beatriz Calvo-Flores Guzman, presented at the 20th Annual WORLDSymposium discussing preclinical data demonstrating that our clinical-stage GCase regulator provided neuroprotection & restored motor function in Parkinson’s disease models following delayed administration.

- Details
- Written by: Developer SM
- Category: Perspectives
- Hits: 292

